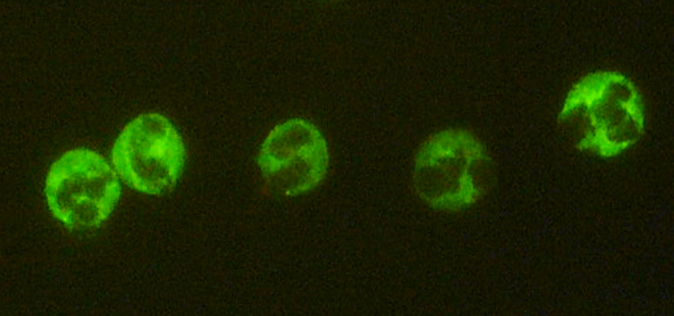
4060 – AKLIDES® cANCA

Highlights
- Slides coated with ethanol-fixed human granulocytes
- Qualitative determination of IgG antibodies against neutrophil cytoplasmic antigens (ANCA)
- Screening test to support the diagnosis of systemic vasculitis (SV)
- Ready-to-use reagents (except PBS buffer)
- Designed for manual processing and automated evaluation using the AKLIDES® or akiron® systems
- High diagnostic sensitivity and specificity for cANCA
- Clear differentiation of cANCA, pANCA, and ANA patterns
- CE marked
Intened Use
The AKLIDES® cANCA is an immunofluorescence assay (IFA) that qualitatively detects IgG antibodies against neutrophil cytoplasmic antigens (ANCA) in human serum using ethanol-fixed granulocytes. It aids in diagnosing systemic vasculitis (SV) alongside other clinical and laboratory findings. The assay supports both manual processing and automated evaluation with the AKLIDES® or akiron® systems and is intended for professional in vitro diagnostic use.
Diagnostic Relevance
Inflammation of various blood vessel walls drives the pathogenesis of systemic vasculitis (SV) and produces characteristic morphological changes. Both arteries and veins can become affected simultaneously. Patients typically present with general symptoms such as exhaustion, fever, and weight loss. The disease course varies depending on which types of vessels are involved.
Anti-neutrophil cytoplasmic antibodies (ANCA) play an essential role in the serological diagnosis of SV. Laboratories usually determine these antibodies by performing indirect immunofluorescence on ethanol-fixed human neutrophils. Technicians distinguish cytoplasmic ANCA (cANCA) from perinuclear ANCA (pANCA) based on their immunofluorescence patterns. Ethanol fixation destroys granular membranes in the neutrophil cytoplasm, allowing positively charged proteins to move to the negatively charged nucleus, which forms the basis for the ANCA screening substrate. Analysts identify cANCA antibodies by their cytoplasmic fluorescence pattern, while they differentiate pANCA antibodies from antibodies to nuclear antigens (ANA) that also display a (peri)nuclear pattern.
Product Specifications
| Title | AKLIDES® cANCA |
| Product code | 4060 |
| Indication | Systemic vasculitis |
| Description | Indirect immunofluorescence assay for the determination of IgG antibodies in human serum against neutrophil cytoplasmic antigens (ANCA) |
| Format | Slides coated with ethanol fixed human granulocytes |
| Incubation time | 60 min. |
| Sample volume | 10 µL serum |
| No. of determinations | 60 (10 x 6) |
Free downloads
SDS [REF 4060][eng] SDS [REF 4060][deu] Flyer [ANCA IFA Overview][eng] Flyer [ANCA IFA Overview][deu]Restricted downloads - Password required
Current version of the instructions for use. The respective valid version for processing the test can be found in the product packaging.