AI-Based ANA HEp-2 IFA Interpretation: Clinical Validation in Routine Diagnostics
Antinuclear antibody (ANA) testing by indirect immunofluorescence assay (IFA) on HEp-2 cells remains the gold standard screening method for systemic autoimmune rheumatic diseases (SARD), including systemic lupus erythematosus, systemic sclerosis and Sjögren’s syndrome1. However, manual interpretation of ANA HEp-2 IFA patterns is time-consuming, operator-dependent and subject to inter-observer variability2.

A recent large-scale study published in Clinica Chimica Acta evaluated whether AI-based ANA HEp-2 IFA interpretation can match the performance of experienced immunologists under real-world laboratory conditions3. The investigation included 2,671 consecutive serum samples analyzed in a routine diagnostic laboratory and compared expert visual reading with automated image interpretation using the akiron® NEO. This analysis is based on the solid results already emerging from a pilot study that had previously highlighted the potential of artificial intelligence in supporting ANA diagnostics4.
Real-World Performance of Automated ANA Diagnostics
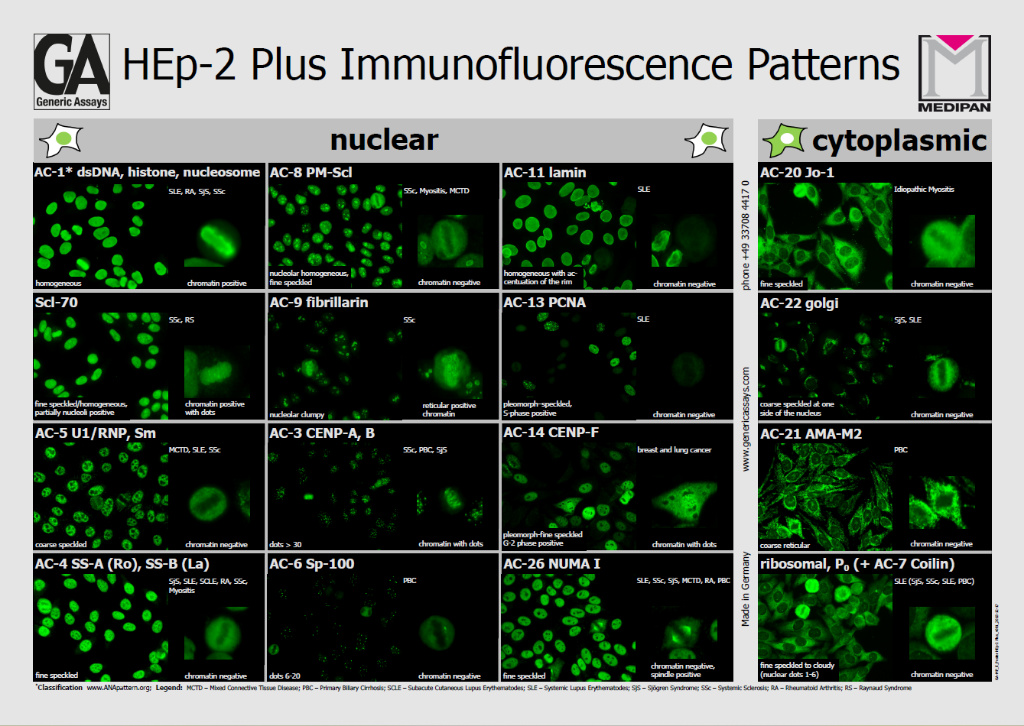
The study assessed ANA positivity/negativity discrimination, titer assignment, and fluorescence pattern recognition according to the International Consensus on ANA Patterns (ICAP) at the competent level.
A good concordance was found for positive/negative ANA classification (κ 0.616–0.699). Agreement improved further when weakly positive samples were excluded.
Pattern recognition performance was similarly robust. Nuclear patterns reached kappa values up to 0.650 in clinically significant titers (≥1:320), while mitotic pattern identification achieved even higher agreement (up to κ = 0.736). Cytoplasmic patterns showed slightly lower concordance, consistent with the inherent variability described in ANA diagnostics literature5.
The results demonstrate that agreement between AI-supported analysis and human observers was within the same range as inter-observer agreement between experienced immunologists, confirming comparable diagnostic performance. This finding indicates that variability in ANA interpretation reflects intrinsic methodological complexity rather than technological limitation.
The automated system showed a slightly lower overall positivity rate compared with one human observer, suggesting a more conservative approach in borderline cases. In routine laboratories, especially outside specialized rheumatology settings, such standardization may help reduce unnecessary follow-up testing triggered by unspecific weakly positive results.
Deep Learning Architecture for ANA Pattern Recognition
The akiron® NEO platform applies hierarchical convolutional neural networks (CNNs) trained for specific analytical tasks, including cell cycle phase detection, nuclear and cytoplasmic pattern recognition, and mitotic analysis. DAPI counterstaining supports accurate metaphase identification, which is essential for ICAP-compliant classification. The system integrates these outputs to generate standardized ANA HEp-2 IFA interpretations aligned with ICAP nomenclature.
Clinical Correlation
In a subset of samples with reflex immunoblot testing, AI-based pattern recognition demonstrated expected serological associations. For example, all AC-3 centromere patterns corresponded to anti-CENP-B positivity, while speckled patterns showed associations with anti-Ro52, SS-A, SS-B, Sm and U1-nRNP antibodies. These findings support the clinical validity of AI-driven ANA classification.
Conclusion
AI-based ANA HEp-2 IFA interpretation demonstrates diagnostic performance comparable to experienced immunologists in large-scale routine practice. While expert review remains essential, automated ANA diagnostics enhance workflow efficiency, improve reproducibility, and reduce subjectivity in ICAP-based pattern classification.
For laboratories seeking standardized, scalable and high-throughput ANA testing, AI-supported HEp-2 IFA analysis with akiron® NEO represents a clinically validated and scalable solution for standardized ANA HEp-2 diagnostics.
Product used in the study
| 4450 – akiron® NEO | IFA analyzer with AI-based software used for automatic ANA, ANCA, nDNA and CytoBead® pattern recognition and intensity/titer assessment |
| 4063, 4065 – AKLIDES® ANA plus | HEp-2 cells based IFA for the determination of IgG antibodies against nuclear and cytoplasmic antigens (ANA) |
Discover the full range of the immunofluorescence products
References
- Bossuyt et al., (2020) Understanding and interpreting antinuclear antibody tests in systemic rheumatic diseases ↩︎
- Rigon et al., (2017) The inter-observer reading variability in anti-nuclear antibodies indirect (ANA) immunofluorescence test: a multicenter evaluation and a review of the literature ↩︎
- Schmidt et al., (2025) Comparison of manual with AI-aided interpretation of ANA HEp-2 IIF assay patterns in a clinical diagnostics lab ↩︎
- Mayr et al., (2025) Pilot Study of AI-Assisted ANA Immunofluorescence Reading—Comparison with Classical Visual Interpretation ↩︎
- Rigon et al., (2017) The inter-observer reading variability in anti-nuclear antibodies indirect (ANA) immunofluorescence test: a multicenter evaluation and a review of the literature ↩︎