4123 – AKLIDES® Anti-GBM

Highlights
- Use of purified rabbit IgG as solid-phase antigen
- Monkey kidney tissue sections as substrate for reliable visualization of antibody binding
- Supports diagnosis of autoimmune glomerulonephritides, including Goodpasture syndrome
- Facilitates differential diagnosis of rapidly progressive glomerulonephritis (RPGN) alongside ANCA and ANA testing
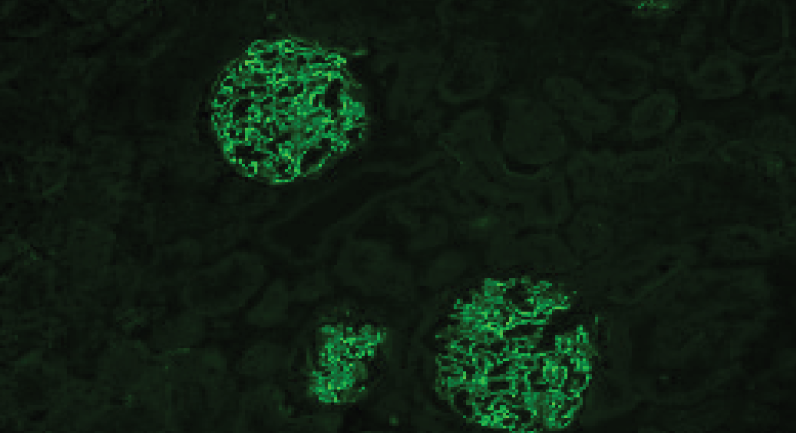
- Characteristic fluorescence patterns enable confident interpretation
- Ready-to-use reagents (except wash buffer) for streamlined laboratory workflow
- Characteristic fluorescence patterns enable confident interpretation
- automated imaging with AKLIDES® and or akiron® systems
- CE marked
Intended Use
The AKLIDES® Anti-GBM is an immunofluorescence assay (IFA) for the qualitative determination of IgG antibodies in human serum against glomerular basement membrane antigens on monkey kidney tissue sections. The AKLIDES® Anti-GBM is intended as an aid in the diagnosis of autoimmune glomerulonephritides in conjunction with other clinical and laboratory findings. The immunoassay is designed for manual processing and automated imaging using the AKLIDES® or akiron® system. The immunoassay is designed for professional in vitro diagnostic use.
Diagnostic Relevance
Goodpasture syndrome is a rare autoimmune disorder in which proliferative glomerulonephritis and pulmonary haemorrhage occur simultaneously. Central to its diagnosis is the detection of circulating autoantibodies targeting the glomerular basement membrane. The GBM acts as a vital barrier between the kidney’s epithelial cells and the underlying connective tissue and plays a key role in ultrafiltration. Rapidly progressive glomerulonephritis often occurs in various autoimmune kidney diseases, making timely and accurate antibody detection essential.
Diagnosis typically requires assessment of multiple antibody types. Anti-GBM antibodies are highly specific for Goodpasture syndrome, while anti-neutrophil cytoplasmic antibodies indicate conditions such as Wegener’s granulomatosis or vasculitis-associated RPGN. Anti-nuclear antibodies are associated with lupus nephritis and other systemic autoimmune diseases. Early detection of these pathogenic antibodies allows clinicians to start immunosuppressive therapy and plasma exchange treatments promptly, which can significantly improve patient outcomes and prevent irreversible kidney damage.
The AKLIDES® Anti-GBM assay provides a sensitive and reliable method for detecting these antibodies. It produces clear fluorescent patterns, enabling precise interpretation and consistent results across routine laboratory workflows.
Product Specifications
| Title | AKLIDES® Anti-GBM |
| Product code | 4123 |
| Indication | Autoimmune glomerulonephritides |
| Description | Indirect immunofluorescence assay for the determination of IgG antibodies against the glomerular basal membrane (GBM) in human serum |
| Format | Slides coated with cryostat tissue sections of monkey kidney |
| Incubation time | 60 min. |
| Sample volume | 50 µL serum |
| No. of determinations | 48 (12 x 4) |
Free downloads
SDS [REF 4123][deu] SDS [REF 4123][deu]Restricted downloads - Password required
Current version of the instructions for use. The respective valid version for processing the test can be found in the product packaging.